 People who are lactose-intolerant lack this enzyme. Lactose is hydrolyzed to glucose and galactose in the intestine by the enzyme lactase. Lactose is a reducing sugar the β -D-glucose residue has a free anomeric carbon, and therefore the glucose residue is on the reducing end. The correct specification of the configuration of the anomeric carbon is critical: an α (1 →4) linkage is not the same thing as a β (1 →4) linkage. The bond is designated a β (1 →4) bond, indicating the configuration of the anomeric carbon ( β ), the number of the anomeric carbon (1), and the number of the carbon (of the second sugar) to which it is linked (4). The anomeric carbon of the β -D-galactose molecule reacts with the C-4 hydroxyl group of the β -D-glucose molecule to form the glycosidic bond (see Figure 1). Lactose is formed from monosaccharides β -D- galactose and β -D- glucose. These carbohydrates are represented and their formulas are written from nonreducing end to reducing end.Ī common disaccharide is lactose, which is found only in milk. In general, disaccharides and polysaccharides contain both reducing and nonreducing sugars. Anomeric carbons involved in glycosidic bonds are nonreducing. Sugars with free anomeric carbons can reduce ferric (Fe 3+) and cupric (Cu 2+) ions, and are called reducing sugars. Glycosidic bonds form when the anomeric carbon of one sugar reacts with a hydroxyl group belonging to a second sugar. 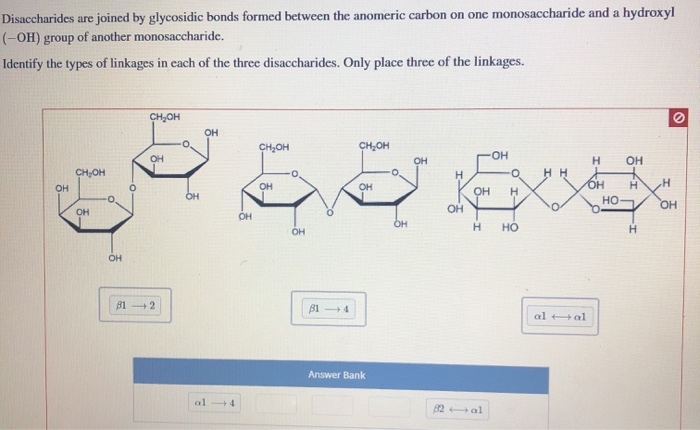
Disaccharides are carbohydrates containing two monosaccharides linked by a glycosidic bond. Although the term "sugar" is commonly used to refer to sucrose, sucrose is only one of a large group of sugars. Sucrose, or table sugar, is the most common disaccharide. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
